Electron microscopy

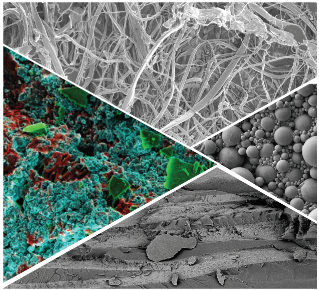
I lead the electron-microscopy effort at the School of Physics & Astronomy at The University of Edinburgh. We have a JEOL JSM-6010, which has a low-vacuum mode and can be equipped with a cool stage. We also have a Zeiss Crossbeam 550 cryo-FIB-SEM-EDX, which can image soft and biological samples in their near-native state at high resolution including soft materials, industrial formulations, and biological matter. For more information and to use the instrument please visit the instrument website and contact the Instrument Scientist Fraiser Laidlaw.
In electron microscopy, electrons rather than light are used for imaging samples. This allows for higher resolution, but typically only the sample surface can be imaged and the sample has to be imaged under vacuum i.e. not in solution. The JEOL Scanning Electron Microscope (SEM) we have access to can image secondary electrons (for a topographical view as in the top image) or backscattered electrons (for a compositional view). The Zeiss cryo-SEM allows imaging of flash-frozen samples, providing a snapshot of soft and biological samples in their near-native state. The Focussed Ion Beam (FIB) enables the controlled milling away of thin slices of the sample. Between each slice an SEM image is taken automatically, thus generating a stack of images that can be reconstructed to obtain a 3D image. The EDX system and a backscatter detector provide rapid elemental mapping. The cryo preparation system can be used to slush freeze, freeze-fracture, sublime and sputter coat samples before transferring to the main SEM chamber, all under cryo conditions; there is also a high-pressure freezer, for example allowing relatively thick samples to be frozen for cryo-FIB-SEM.
Confocal microscopy
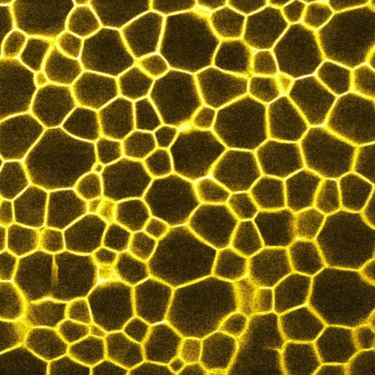
I have nearly two decades experience of confocal microscopy, which can be used to characterize the 3D microstructure of (typically) fluorescently labelled samples in-situ and in real time at an optimal resolution of 150 nm lateral and 500 nm axial. As it uses laser light to excite the fluorescent dye and then records the emission light, it works best on samples that can be refractive-index matched i.e. those in which all components of the sample have the same refractive index. All the same, useful information can also be obtained from opaque samples, e.g. about the microscopic structure at the edge of the sample.
We have access to a Zeiss LSM700 confocal microscope and a Leica TCS SP8 tandem scanner, the latter can image at 25 fps.
Digital photography

I have nearly two decades of experience in scientific digital photography. Visual observation can be surprisingly useful when characterizing soft materials, as the building blocks are of order 1 micron, which means they interact strongly with light, and so it often reveals useful information about the sample quickly. For example, in this photograph, the Bragg colours under white-light illumination suggest that the sample consists of sub-micron spheres arranged in a crystalline packing; the dark stripes suggest crystal twinning.
For visual observations, we use a range of equipment including Raspberry Pi operated webcams, USB cameras, and digital SLR cameras.
Image analysis

Microscopy and photography lead to images of soft materials that are often beautiful in their own right, but they also contain a wealth of quantitative information that can be extracted using image analysis. I have nearly two decades of experience in scientific image analysis, from quantifying crystal quality from SEM images to extracting shear strain from confocal-microscopy time series. In recent years, I have also started exploring machine-learning approaches in image analysis. In 2023/24, I have launched a new undergraduate course called Scientific Image Analysis.
Though trained in IDL, I now mostly employ Fiji (Image) and Python for image analysis, particularly scikit-image and numpy.
(Interfacial) rheology
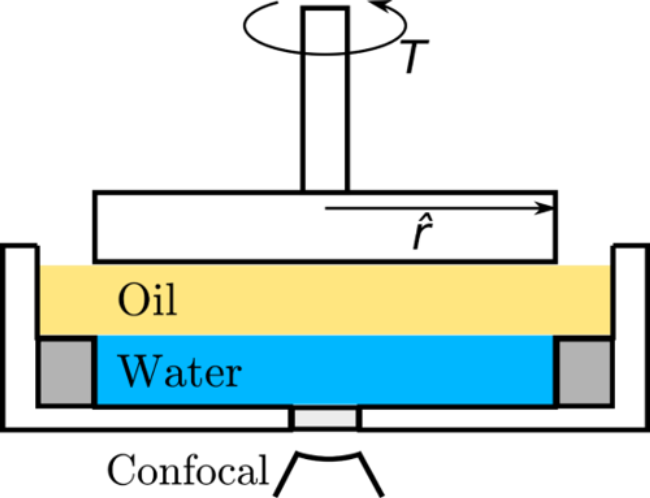
Rheology is the science of the deformation of materials. Soft matter often has solid or liquid-like properties, depending on the timescale probed. For example, think of Silly Putty: it bounces back if you through it at a wall (displaying elastic, solid-like behaviour at short timescales), but it takes the shape of the container if you leave it alone (displaying viscous, liquid-like behaviour at long timescales). Hence, it is no surprise that soft matter physicists often study the rheology of soft materials. I have a decade of experience in rheology and interfacial rheology. The latter is the study of the mechanical properties of interfaces, which contributes for example to our understanding of the stability ('shelf life') of emulsions e.g. vinaigrette, milk and paint. Recently, I led a project that was published and featured in the Journal of Rheology, entitled Contactless interfacial rheology: Probing shear at liquid–liquid interfaces without an interfacial geometry via fluorescence microscopy, see also the schematic and the corresponding Scilight.
Dynamic (DLS) and Static Light Scattering (SLS)
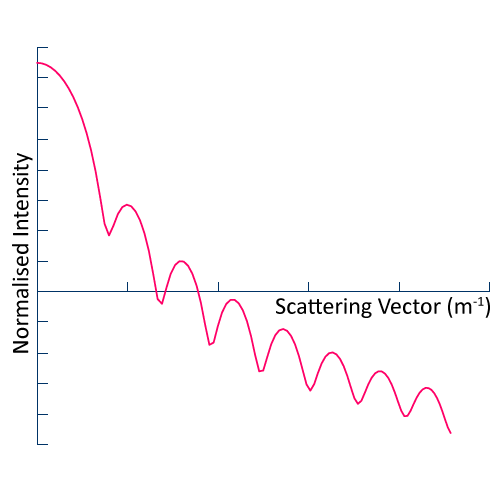
In Static Light Scattering (SLS), we measure the scattered intensity as a function of scattering angle for a suspension of colloidal particles i.e. liquid or solid particles suspended in a liquid (see figure). By fitting the data to an appropriate model for Mie scattering, we obtain the average particle size and variance therein (particle size range 100 to 2000 nm). SLS can also be used to determine molecular weight.
In Dynamic Light Scattering (DLS), we measure the fluctuations of intensity of speckles in scattered light, which we then convert into a decorrelation curve. By fitting the curve to an appropriate model, we can extact quantitative information of the system, for example the average particle size.
We usually employ an ALV LSE-5004 based set-up, which allows for temperature controlled measurements in which the scattering angle can (practically) be varied between 30 and 150 degrees.
X-ray micro-CT

In X-ray micro-CT (CT), we essentially take X-ray transmission images of a sample at various angles covering a full 360 degrees rotation. The resulting images are then analyzed by a computer program that can reconstruct them into a 3D image stack: a series of 2D images at various heights throughout the sample. One advantage of X-rays is that most materials are relatively transparent to X-rays, so full 3D reconstructions of visually opaque materials can be made.
In recent years, I have also supervised projects on machine-learning approaches to extract quantitative information from total-body X-ray CT and PET medical imaging data sets.
Potentiostat / galvanostat
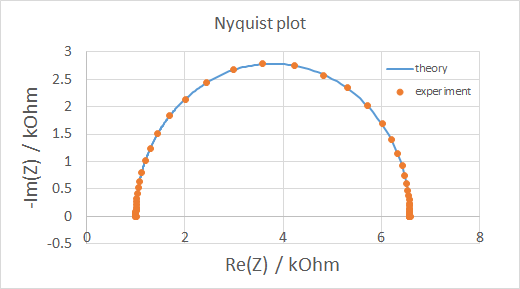
Some soft materials have promising properties for energy applications, for example bijels and bijel-like structures have been used as scaffolds for enhanced battery electrodes and hybrid electrolytes. A potentiostat / galvanostat is used for electrochemical charcaterization of samples. Simply speaking, either the voltage across (potentiostat) or the current through (galvanostat) the sample is controlled, while the current/voltage is measured. We mostly use this machine for cyclic voltammetry (essentially generating I-V curves) and electrochemical impedance spectroscopy (plotting imaginary vs real part of impedance).
We usually employ a VersaSTAT 3 from Princeton Applied Research, supplied by Blue Scientific (UK).